
How Proper Crop Protection Ensures Global Food Safety and Helps the Planet

In recent decades, a wide variety of crop protection products have provided farmers with a set of carefully tailored tools for the production of safe and healthy crops. SBDIGIT / Getty Images
By J. Erik Fyrwald
World Food Safety Day earlier this week reminded us of a harsh reality: Roughly 600 million people around the world suffer from food-borne illnesses, while an estimated 3 million die from illnesses contracted from unsafe food or water.
This annual toll touches every part of the world, though the worst impacts are felt in regions like sub-Saharan Africa and South Asia, where there is less access to refrigeration and other modern food-preservation technologies. While major improvements have been achieved in some regions since the 1980s, the problem is still vast and urgent.
Dangers in food supply can occur at any stage of the farm-to-fork process. Many of these relate to food preparation, and every cook must take all available precautions to maintain a clean, safe kitchen. Less well known is what can be done to ensure food safety earlier in the process, beginning on the farms where it is produced.
Agronomists Have an Outsized Role to Play
In recent decades, thanks to successful research and development in the field of agronomy, a wide variety of crop protection products have provided farmers with a set of carefully tailored tools for the production of safe and healthy crops.
For example, Fusarium is a common fungus that can produce deadly mycotoxins in wheat, corn and barley. Recently developed broad-spectrum fungicides are now being used to prevent the spread in these crops. These agents play an active role in protecting the health of consumers and avoiding waste in having to destroy the infected grains.
In the past, pesticides, herbicides and fungicides have themselves sometimes been viewed as threats to the safety of food and water.
But innovation in crop protection products has come a long way since Rachel Carson pointed out the risk they posed in her seminal 1962 book Silent Spring. Today’s crop protection agents are developed with both human health and the environment firmly in view.
Over the course of the last 60 years, these agents have become more targeted and more effective and they are applied much more sparingly. Since the 1950s, the agronomy sector has achieved a remarkable 95% decrease in the average application rate of active ingredients per hectare. At the same time, multiple innovations have led to crop yields from farmland more than tripling. These measures to make everyone’s food safer have been reinforced with the Codex Alimentarius, the open, transparent system of international standards established in 1963 to protect consumer health and promote fair practice in the food trade.
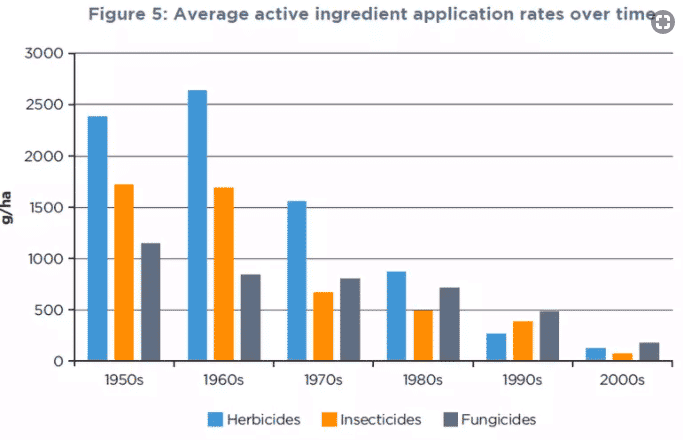
Crop-protection has been used more sparingly over recent decades. Source: Phillips McDougall, 2017
A Deep Respect for Health and the Environment
As it happens, more than a few of today’s crop protection products are obtained from natural sources or are so thoroughly tested by time that they are considered traditional practices. This means they are commonly found in use on farms that specialize in growing organic produce.
Those of us working in the field of agricultural science are well aware these advances are not always appreciated by people who wish insecticides and fungicides were not a part of the global food system. Viewed in a certain light, I agree with them. But I have seen first-hand the destruction wrought by pests and the impact of a failed crop on farmers and their families. The devastation inflicted by swarms of locusts this year in Africa and Asia is just one case in point.
Addressing the Challenge of Climate Change
The need for effective crop protection products has steadily grown as rising global temperatures have expanded the range of many pests and blights. Just last summer in the Netherlands, a farmer told me he was seeing an insect in his fields he had never seen before.
Our team researched it and found the pest was well known in Brazil – Tuta absoluta is a miner fly whose larvae make corridors in the leaves by eating the cells – and becoming a problem in northern climates as a result of warmer winters. One of our goals as an industry is to provide farmers with the safest and best means of keeping threats like these at bay.
When viewed at macroscopic scale, advances in agronomy clearly represent an important part of the solution to climate change. The latest crop protection products address climate change directly – by enabling farmers to produce more food per unit of land than ever before in the history of agriculture. This is critical if we hope to bring a halt to the expansion of human activity into regions such as the Amazon rainforest – a precious natural resource that serves a higher common purpose as a carbon sink and biodiversity preserve than as a cornfield.
Progress has been made on crop protection solutions that help feed humanity safely while protecting the environment, but we should all take a moment on this World Food Safety Day to strengthen our resolve. Even in the face of a global pandemic and myriad other challenges – we must keep moving forward in our efforts to safeguard people and the planet.
Reposted with permission from World Economic Forum.
- Trump Administration Asks Court to Re-Hear Case That Banned ...
- 10 States Report Crop Damage From Illegal Dicamba Use on ...
- Farming Without Pesticides: How Can We Make Agriculture Greener ...
- Environmental Justice Activists React to EU Farm to Fork Strategy
- The Extinction Crisis: Coming to a Dinner Table Near You? - EcoWatch
- Growing Food and Protecting Nature Don’t Have to Conflict – Here’s How They Can Work Together - EcoWatch

 233k
233k  41k
41k  Subscribe
Subscribe 