
Affordable, Self-Administered COVID-19 Tests Could Use Your Cell Phone
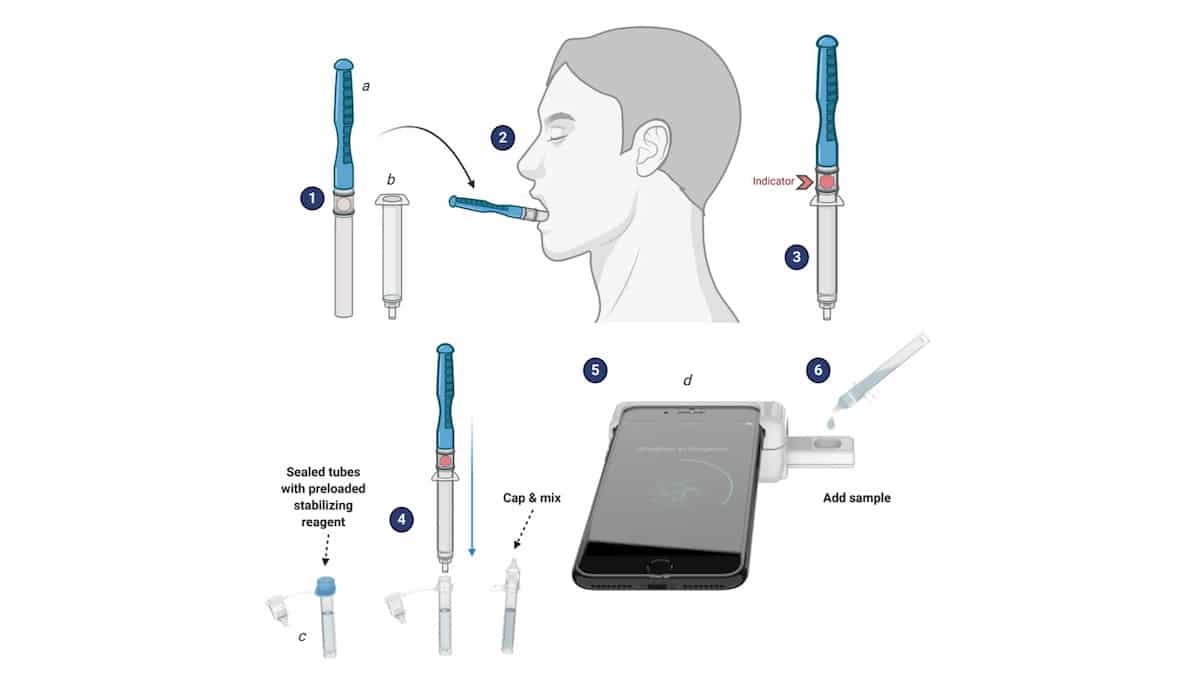
A graphic shows how Rhoel Dinglasan's smartphone-based saliva test works. University of Florida
As the world continues to navigate the line between reopening and maintaining safety protocols to slow the spread of the coronavirus, rapid and accurate diagnostic screening remains critical to control the outbreak. New mobile-phone-based, self-administered COVID-19 tests being developed independently around the world could be a key breakthrough in making testing more widely available, especially in developing nations.
Point-of-care testing (POCT) involves the use of easily portable devices to carry out analytical testing outside of a central laboratory, in places like long-term care facilities, local clinics and patients’ homes, a new study by the National Center for Biomedical Information said. POCT provides early and fast diagnostic results; when combined with microfluidic systems that can measure traces of diseases and infections based on small amounts of fluids with daily high sensitivity and speed, POCT can result in faster, more cost-effective diagnostic instruments that do not require any trained healthcare workers to administer them, the study reported. Smartphone-based microfluidic POCT uses personal mobile devices to carry out the whole analytical process using bodily fluids like saliva. Critically, this also will result in earlier diagnoses that can help slow the spread of diseases like COVID-19.
Recently, Rhoel Dinglasan, a University of Florida infectious diseases professor and his industry collaborators won second place in a national technology competition sponsored by the National Institutes of Health for a smartphone-based, rapid saliva test they developed that can be used to diagnose COVID-19, along with two other diseases of major global importance, malaria and anemia. The test adapted the team’s existing mobile-based test for malaria to also test for anemia and COVID-19, explained Dinglasan in a University of Florida Health news release.
“This test will leverage existing technology to fill a pressing and unmet need in global public health by allowing us to differentiate between three diseases that can present similarly in terms of symptoms,” Dinglasan said in the release. “As treatment and containment protocols differ for each disease, the proper diagnosis delivered in literally minutes could save lives.”
Their solution is the CLIP-CAM, an adapter that attaches to a smartphone that fits a detection cassette, the release detailed. A saliva sample is self-collected and activated, and a droplet is put into the cassette. The camera flash is used to excite the detection system in the cassette, which can then be read by a smartphone app. Results are available in 15 minutes or less, Dinglasan said, the release reported.
He further explained the importance of quick diagnoses to the Gainesville Sun: “When we go out to work in these different countries, we need to differentiate when a person comes in, we need to know if it is COVID-19, malaria or anemia. Without that ability, how would we know if we should send this person home to quarantine because we think he has COVID-19? What if it is malaria? You need to give that child or adult anti-malaria drugs or they will die.”
Similar prototyping is underway around the world. At the University of Utah, electrical and computer engineering professor Massood Tabib-Azar adapted the platform he used to detect the Zika virus to now detect COVID-19, reported BioSpace. His sensor can also test for the presence of the virus on surfaces like door handles or desks by brushing a swab on a surface and then onto the sensor, the news report said. Tabib-Azar is also exploring the potential of detecting COVID-19 in floating microscopic particles in enclosed spaces like an elevator.
The Utah sensor is reusable, because samples can be destroyed using a small electrical current to disintegrate viruses or saliva can be placed on a disposable sheet like a piece of paper put on top of the sensor, Tabib-Azar told BioSpace. His test is also designed to upload the results to a central server that can map out positive results in an area to give public health experts a better idea of where outbreak hotspots are, the report said.
“Mobile-based epidemic monitoring is nothing but a logical next step, because only the mobile devices that move with people can keep up with the contacts they make,” Tabib-Azar said, speaking of related research in IEEE Access.
In Ontario, Canada, a third diagnostic prototype called Atlas hopes to use the same saliva-to-mobile-phone testing concept to make testing more widely available, from remote communities to front-line screening, reported HospiMedica.com. Atlas is designed as a single-use, disposable device that could deliver results in 20 minutes.
All three tests have received enough funding to get through the prototype phase. Additional support will help to quickly transform the design concepts into mass-produced products for global health.
- FDA Approves First In-Home Test for Coronavirus - EcoWatch
- Trump Plans to End Federal Funding for COVID-19 Testing Sites ...

 233k
233k  41k
41k  Subscribe
Subscribe 